Introduction
Thanks for tuning in to Mastering the Microbiome! The goal of this newsletter is to feature exciting new research that is unraveling the mysteries of the microbiome and paving the way for clinical advances.
If you haven’t already, you can subscribe to Mastering the Microbiome, or share it with friends and colleagues:
For many years, we have been fighting off infections by boosting the microbiome.
The classic case is C. difficile infection. C. difficile tends to afflict people in hospitals or long-term care homes. This often means older adults, but people of all ages are at risk, especially those who are immunocompromised. The typical treatment to clear the infection is a round of antibiotics – but about 1 in 6 people who acquire these infections are re-infected again soon after antibiotic treatment. These recurrent C. difficile infections (CDI) cause persistent diarrhea, abdominal pain, fever, and other symptoms.
For those who experience recurrent CDI, it is a case of the cure being worse than the disease. Antibiotic treatment severely damages the microbiome, and as a consequence, makes some patients even more susceptible to infection. Why only some individuals respond in such a negative way - and others clear the initial C. difficile infection successfully without recurrence, is still an open question. But it is likely that the initial state of the microbiome (how likely it is to bounce back after antibiotic treatment), the immune system, and other factors play a role.
What is not in doubt is the usefulness of fecal microbiota transplant (FMT). FMT has been used, quite successfully, to treat recurrent CDI infections. The cure rate is around 90%, a remarkable figure for any kind of therapeutic. FMT is now supported by a large medical infrastructure – including donor stool banks at local hospitals and large external stool banks run by companies like OpenBiome. Although FMT has been around a long time – since 1983 for CDI, as far back as 1958 for treatment of colitis, and going back millennia as recorded in ancient Chinese medical texts from the fourth century – the first FDA approved FMT drug (REBYOTA) came only in 2022. In 2023, the FDA approved the first oral FMT capsule under the brand name Vowst.
FMT has long been delivered primarily through colonoscopy or enema, which are more cumbersome procedures to say the least. The fact that oral FMT delivery has lagged behind reflects reasonable questions that it could be as effective and safe as the more direct modes of delivery through the back end of the digestive tract. Recent studies, however, suggest that it can be.
Whatever the mode of delivery, The basic idea of FMT is that you are taking the microbiome from a healthy donor and introducing it to the body of someone sick with CDI. As the donor microbiome establishes itself, it creates conditions that help fight off the infection.
Here’s a nice graphic from Knowable Magazine that outlines the whole process:
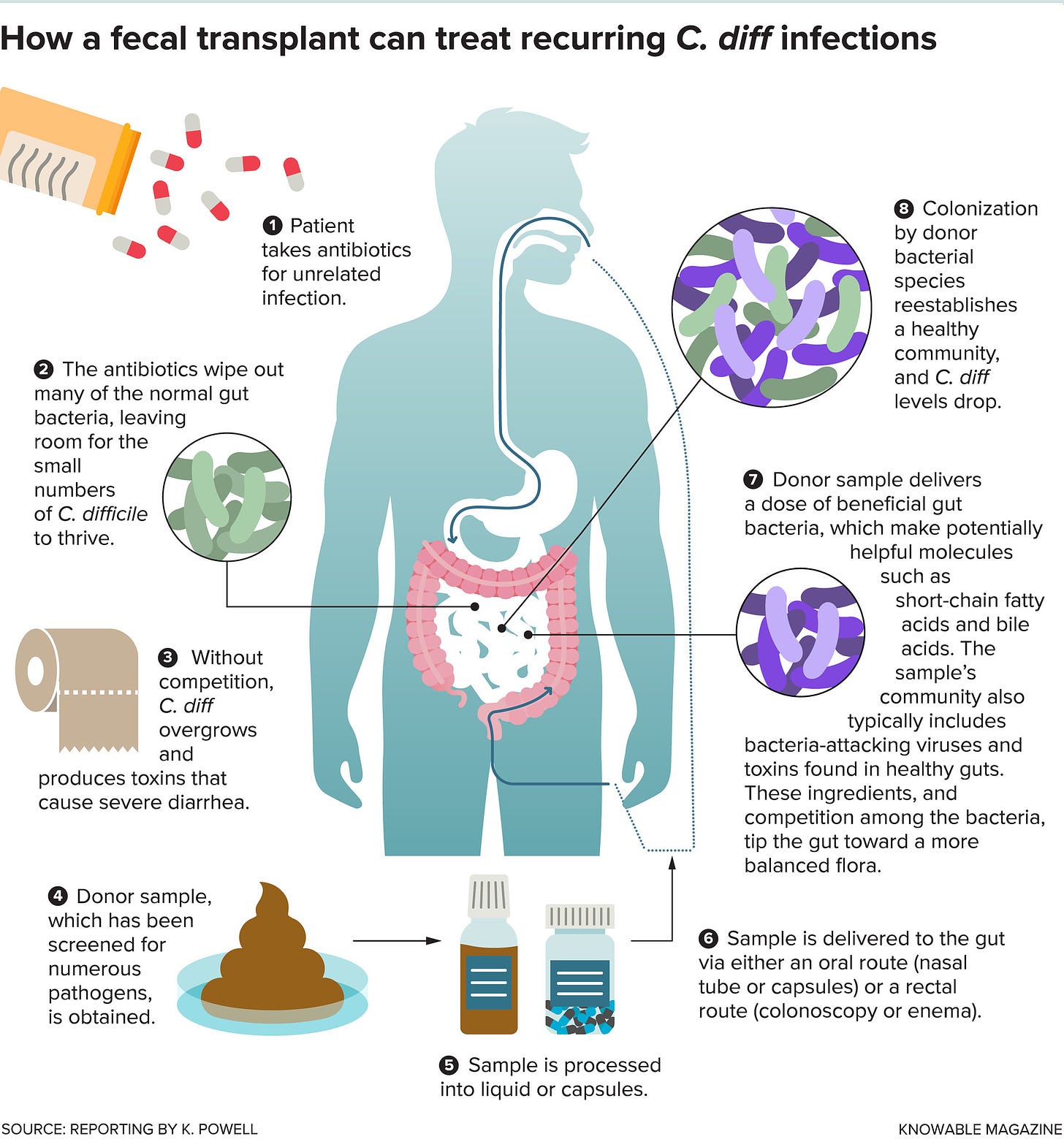
Why does this work? The concept of colonization resistance – that the microbiome can act as a barrier to entry for infectious pathogens – is an active area of research. There are multiple possible defense mechanisms that scientists now recognize. Members of the microbiome can produce compounds that are toxic to the invading pathogen, consume nutrients that the pathogen needs to survive, physically contact and combat the pathogen, or stimulate the immune system to display more robust anti-pathogen activity. There are also potentially other mechanisms of action that we are not yet considering.
In short, antibiotics kill off the healthy defensive microbiome. Administering FMT is like calling in reinforcements.
But are there better alternatives? Despite its great success, there may be easier and even more effective ways to treat (or better yet prevent) recurrent CDI and other secondary infections.
There have been some successful alternative efforts thus far. The combination of the Lifeway Kefir smoothie (which contains a number of probiotic species) with antibiotics has shown success in treating recurrent CDI. Notably, the 26 patients in this study were all given the option to go on FMT and all chose this alternative antibiotic-smoothie treatment - many citing the expensiveness and complicated nature of the FMT procedure.
More recently, researchers have explored the use of so-called ‘next-generation probiotics’ as a more targeted treatment strategy. If you think about it, taking the microbiome from a healthy donor and transplanting it into a patient with CDI is a pretty heavy-handed approach, and prone to poor engraftment if the recipient’s body is not hospitable to the donor’s fecal microbiome (another area of active research - why do some people respond more poorly to FMT than others). FMT clearly works, but it may be more effective to isolate particular bacteria in the healthy microbiome that are contributing to the success of FMT and administer them individually.
Next-gen probiotics, by one definition, are simply probiotic bacteria that have not been used extensively to date (i.e., not the common Bifidobacterium and Lactobacillus probiotics that have been in use for many years). Prospective next-gen probiotics have been identified through metagenomic sequencing studies of the microbiome that indicate which species are associated with a healthy clinical condition (or depleted in people with disease).
Ideally, a next-gen probiotic candidate would be administered at the same time as antibiotics to treat C. difficile infection. This is a preventative measure. You are not waiting to see if someone develops recurrent CDI – you are giving them a probiotic to prevent that from happening altogether. In addition, you want to make sure that your probiotic is itself resistant to the antibiotic treatment and won’t simply be killed off before it can have any beneficial effects.
To this end, scientists have tested individual species in the lab that are resistant to antibiotics used to treat CDI and can therefore be administered successfully as an adjunct probiotic therapy. For example: L. reuteri. L. reuteri is of interest to the biomedical research community because it produces the compound reuterin, which is known to be toxic to C. difficile. Scientists from the Baylor College of Medicine, Texas Children’s Hospital, and the University of Texas Medical Branch found that to successfully kill C. difficile in a laboratory culture, you need to treat simultaneously with both L. reuteri and glycerol, the precursor chemical compound that L. reuteri utilizes to produce reuterin.
Though the reuterin paper is old news at this point – it came out in 2017 – there are many aspects of the story that are still very much worth discussing. For one, L. reuteri has now been involved in nearly 200 clinical trials for various clinical purposes – 25 of which have recently started and/or are seeking participants. This demonstrates the potential value of individual probiotic species as therapeutic agents.
Another important point is that the success of a probiotic, or FMT for that matter, is clearly dependent on context. If a key way that the microbiome resists invasive pathogens is by producing compounds that are toxic to those pathogens, and the microbiome needs needs certain resources to produce these defensive compounds, then microbiome-based therapies (FMT, individual probiotics, and probiotic consortia) are unlikely to succeed if the right resources are not present in the right amounts.
Perhaps a deficiency in glycerol (or some other necessary substrate) explains why FMT fails in some patients. Dietary deficiency may help explain initial infection risk as well. For example, studies have shown that diets involving caloric restriction can disrupt the microbiome’s capacity for colonization resistance. Along the same lines, taking certain dietary supplements could help prevent or fight off infection by boosting the natural defense capacity of the microbiome.
None of these are new ideas – but they are all still very active topics of research and potential hotbeds for novel discovery. We still have a very limited sense of how diet and the microbiome interact to promote colonization resistance, and there will surely be many new studies on this topic to be featured in future editions of this newsletter.


I contacted c-diff after being in the hospital for a week, I had it for over 6 months and felt like I was on the brink of death. Several rounds of antibiotics didn’t help. I was desperate and decided on FMT, against 4 doctors advisement, they said it wasn’t FDA approved. (Hmm kinda like the vaccines.) After 2 days of transplant I knew I was going to be ok. FMT saved my life.